
Medical technology companies operate in one of the most complex regulatory environments in the world. From device development through post-market surveillance, regulatory teams must manage global registrations, documentation, and regulatory commitments across multiple markets.
This is why medical device regulatory information management has become essential for modern MedTech organizations.
Regulatory Information Management (RIM) software provides the digital infrastructure needed to manage regulatory complexity, maintain compliance, and accelerate product approvals worldwide.
Many MedTech companies still rely on spreadsheets, shared drives, and disconnected systems to manage regulatory data. However, as regulatory frameworks such as EU MDR, IVDR, and FDA requirements continue to evolve, centralized regulatory information management platforms are becoming critical for maintaining regulatory oversight and operational efficiency.
What Is Medical Device Regulatory Information Management?
Medical device regulatory information management refers to the processes, systems, and technologies used to organize and control regulatory data across the medical device lifecycle.
A modern RIM platform supports regulatory teams by managing:
- Device registrations and approvals
- Regulatory submissions and timelines
- Technical documentation and design dossiers
- Health authority communications
- Global regulatory requirements
By centralizing regulatory data into one platform, organizations gain a single source of truth for regulatory operations.
This improves visibility across markets and helps regulatory teams manage compliance with greater confidence.
The Growing Regulatory Complexity in the Medical Device Industry
Medical device companies must comply with a wide range of regulatory frameworks, including:
- FDA 510(k), PMA, and De Novo pathways in the United States
- EU MDR and IVDR requirements in Europe
- Country-specific device registrations worldwide
Regulators increasingly require structured regulatory data, traceability, and lifecycle oversight.
Without a system for medical device regulatory information management, organizations risk:
- Missed submission deadlines
- Incomplete regulatory documentation
- Inspection findings
- Delayed product launches
A centralized RIM system helps organizations manage this complexity more effectively.
Key Benefits of Medical Device Regulatory Information Management Systems
Centralized Device Registrations and Regulatory Data
RIM software consolidates regulatory information into one centralized platform.
This allows regulatory teams to:
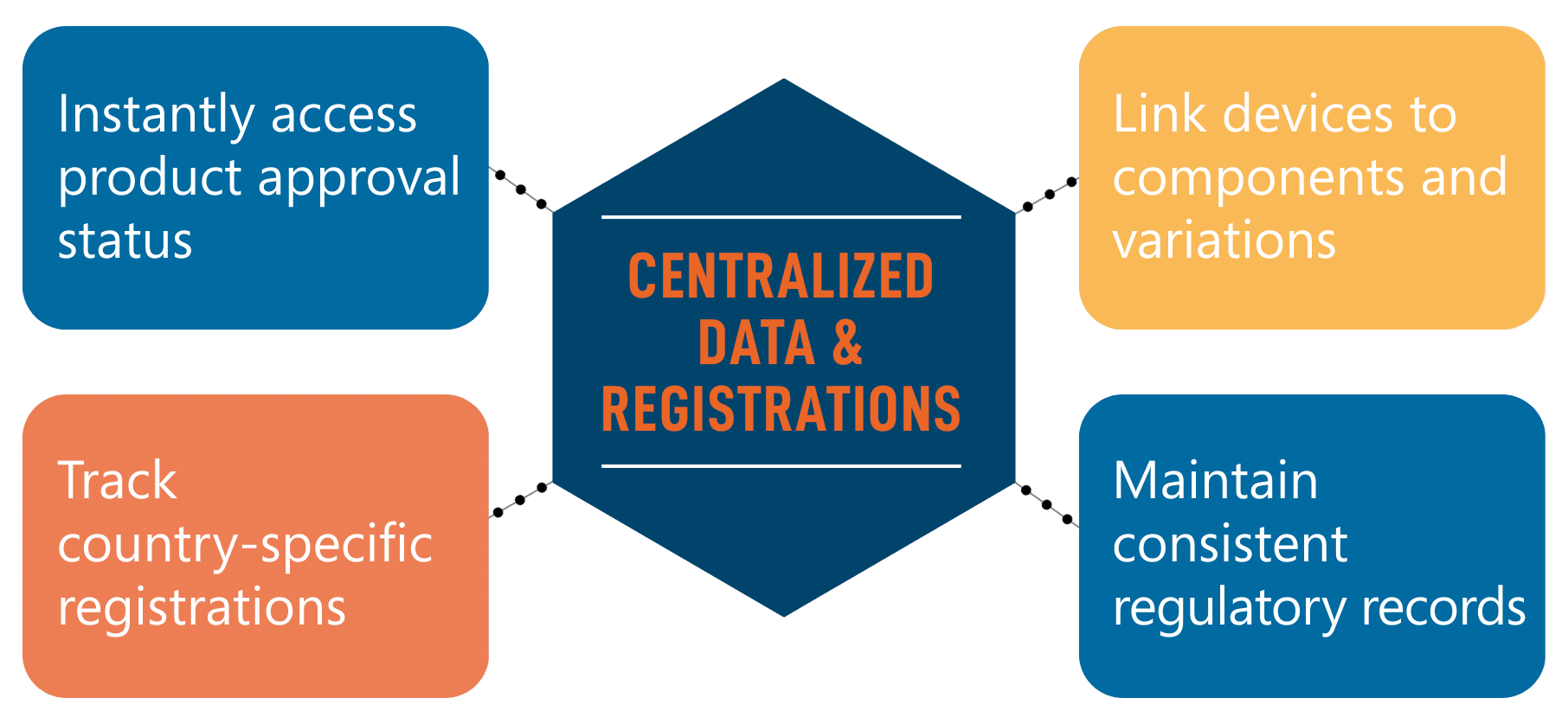
- Instantly access product approval status
- Track country-specific registrations
- Link devices to components and variations
- Maintain consistent regulatory records
This improves visibility and reduces duplication across global regulatory operations.
Improved EU MDR and Global Regulatory Compliance
Improved EU MDR and Global Regulatory Compliance
EU MDR and IVDR have significantly increased regulatory documentation and lifecycle monitoring requirements.
Medical device regulatory information management systems support compliance by managing:
- Technical documentation tracking
- Regulatory variations and change control
- Certificate expiration monitoring
- Post-market surveillance reporting
Automated alerts help ensure that regulatory milestones are never missed.
Structured Submission Planning and Tracking
Manual submission tracking introduces operational risk.
RIM systems provide:
- Structured submission planning
- Deadline monitoring and alerts
- Real-time regulatory pipeline dashboards
- Improved submission transparency for leadership teams
This improves submission efficiency and reduces regulatory delays.
Cross-Functional Collaboration Across Regulatory Teams

A centralized medical device regulatory information management platform improves collaboration by providing shared visibility into regulatory commitments and approvals.
Audit and Inspection Readiness
Health authority inspections require structured regulatory documentation.
RIM software supports inspection readiness through:
- Complete audit trails
- Version-controlled regulatory data
- Centralized documentation access
- Regulatory reporting capabilities
This improves confidence during FDA inspections and notified body audits.
Scalability for Growing MedTech Companies
As device portfolios expand and new markets are entered, regulatory complexity increases. RIM software scales to support:
- Multiple product families
- Expanding global registrations
- Post-market commitments
- Lifecycle management
Growing MedTech companies can avoid costly system replacements by implementing a scalable RIM platform early.
Why Medical Device Regulatory Information Management Is a Strategic Investment
Effective medical device regulatory information management does more than support compliance. It improves operational performance across regulatory teams.
 FASTER TIME TO MARKET
FASTER TIME TO MARKET
 REDUCE REGULATORY RISK
REDUCE REGULATORY RISK
 GREATER EXECUTIVE VISIBILITY INTO REGULATORY STATUS
GREATER EXECUTIVE VISIBILITY INTO REGULATORY STATUS
 DATA-DRIVEN REGULATORY STRATEGY
DATA-DRIVEN REGULATORY STRATEGY
 IMPROVED RESOURCE ALLOCATION
IMPROVED RESOURCE ALLOCATION
In today’s MedTech industry, regulatory performance directly impacts commercial success.
Final Thoughts: What Makes Ennov RIM the Best Choice for Growing Life Sciences Companies?
Growing life sciences companies require more than just basic compliance tools. They need a regulatory platform capable of supporting global operations, complex product portfolios, and evolving regulatory frameworks.
Ennov RIM provides a comprehensive platform for medical device regulatory information management, designed specifically for the needs of life sciences organizations.
Ennov’s regulatory platform helps companies:
- Manage device registrations and submissions globally
- Maintain structured regulatory data across product lifecycles
- Improve collaboration between regulatory, quality, and clinical teams
- Ensure ongoing compliance with evolving regulatory frameworks
Because Ennov RIM is built as part of a broader life sciences quality and regulatory ecosystem, organizations benefit from integrated workflows between regulatory information management, quality management, and clinical operations.
This integrated approach provides:
- Greater regulatory visibility
- Stronger compliance oversight
- Reduced operational silos
- Faster regulatory decision-making
For growing MedTech organizations, Ennov RIM provides the scalability, structure, and visibility required to support long-term regulatory success.


