IDMP Solution
for Data Management and Compliance
IDMP data management software to organise medicinal product data, support IDMP compliance, and operationalise IDMP across your portfolio.
Designed for Regulatory Affairs, Regulatory Operations, and IDMP program owners.
- Centralise IDMP data and standardise product definitions
- Align data with EMA expectations, including PMS readiness and improved data quality
- Maintain a structured IDMP data model for consistent regulatory execution
- Reduce duplication with governed workflows and controlled updates
Simplify IDMP Management
The ISO Identification of Medicinal Products standards are a set of common, global standards for data elements, formats, and terminology to identify and exchange information about medicinal products.
The introduction and progressive implementation of new IDMP standards means that organizations will need to capture and manage significantly more regulatory data than ever before, which represents a significant challenge to the industry as a whole.
Effective data management involves significant effort, is time-consuming and costly. Ennov IDMP and our IDMP services simplify the process of maintaining your IDMP data while transitioning to the new standard.
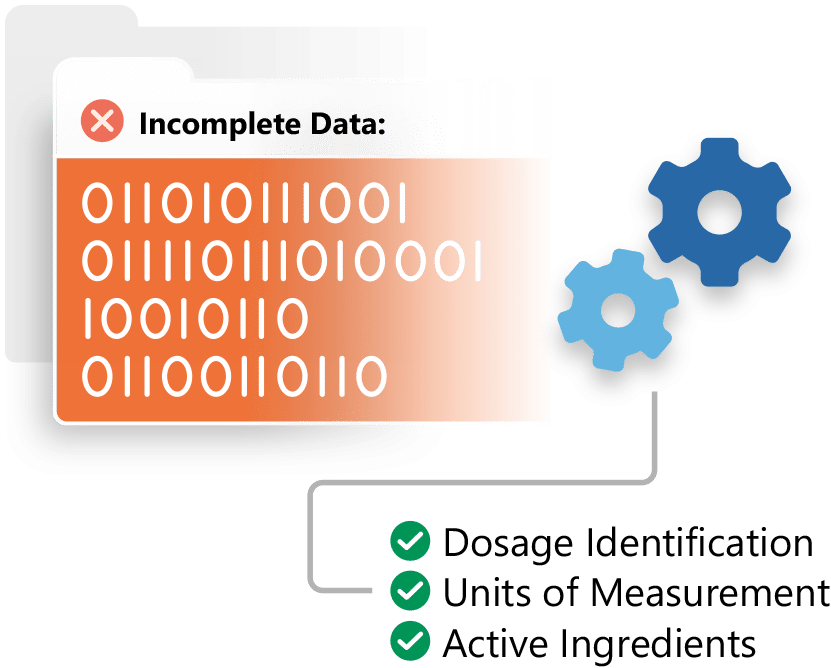
Ensure IDMP Data Quality with Ennov's IDMP Solution
Ennov IDMP is a quality controlled solution for data management in support of the new IDMP standard.
Leverage your existing data to get a jump start on IDMP. Inventory, identify, cleanse and integrate your data, making your organization IDMP ready.
Mine and extract information contained in your regulatory documentation and other enterprise systems to ensure completeness.
Use Our IDMP EASI Calculator to Estimate Your PMS Workload
It’s free, and only takes 5 clicks to discover how much time you can save with our IDMP EASI tool.
Connected to the Future
Ennov RIM is IDMP ready. We currently support the EMA SPOR Referentials lists (controlled vocabularies) within the system and flag each data element with an “IDMP Term” indicator.
As the SPOR data services specifications evolve, they will be systematically incorporated into the solution – taking full advantage of the Ennov Platform’s flexibility and configurability.
Ennov IDMP is a key component in our comprehensive Regulatory solution suite. In combination with Ennov Doc, Ennov RIM, Ennov Dossier, Ennov Report and our REST API, Ennov has the solution today to whatever challenges tomorrow may bring.
Did you know that Ennov supports your IDMP-readiness, even if you use another RIM?
Our agnostic IDMP EASI solution works with any RIM system to ensure IDMP compliance.
Prepare for IDMP Compliance
Ennov recognizes that compliance with ISO IDMP represents a huge challenge for companies and addressing this challenge places an additional burden on their Regulatory resources.
In an effort to help the industry effectively and efficiently meet the requirements of this new standard, Ennov offers an IDMP Readiness Assessment service as a crucial first step in preparing for IDMP submissions.
Performing the assessment results in a documented evaluation that identifies the various data sources (both structured and unstructured) and provides an accounting of the gaps that will need to be addressed to ensure IDMP compliance.
Core Capabilities
- Use of available controlled vocabularies
- Assistance with data extract, cleansing and migration
- Pragmatic IDMP data management
- Time and effort savings
- Increased data quality and integrity
Key Features
- Metadata orientation
- Highly configurable
- Information tracking
- Seamless integration with the Ennov Regulatory Suite
- Robust reporting and analytics
IDMP Software FAQs
What is IDMP software?
IDMP software helps life sciences companies organise and govern Identification of Medicinal Products data in a structured way, so product data is consistent, traceable, and ready for regulatory use. It supports IDMP data management across product definitions, changes, and downstream processes that rely on accurate data.
How does an IDMP solution support IDMP compliance?
An IDMP solution supports IDMP compliance by centralising medicinal product data, applying governance and data quality controls, and maintaining a structured IDMP data model over time. This reduces duplication, improves consistency, and helps teams stay audit-ready as requirements evolve.
Is Ennov’s IDMP compliance software ready for the 2026 deadlines?
Yes. Ennov’s IDMP compliance software is designed to support the June and December 2026 deadlines by helping teams prepare, manage, and maintain structured product data in line with IDMP expectations. It enables ongoing data management and controlled updates, so readiness is not a one-time project.
What does IDMP data management include in practice?
IDMP data management typically includes data inventory, cleansing, mapping to a structured model, enrichment, validation, and ongoing governance. The goal is to make IDMP execution repeatable across products and markets, with clear ownership and traceability.
How does an IDMP system help with IDMP implementation?
An IDMP system supports IDMP implementation by providing a structured data model, workflows for data ownership and updates, and controls that reduce rework. This helps regulatory and data teams coordinate changes and maintain data quality as product information evolves.
What if we are not using Ennov RIM, can we still use Ennov for IDMP?
Yes. If you’re not using Ennov RIM, Ennov offers an agnostic option called IDMP EASI that supports IDMP readiness while working with your existing RIM. It is designed for organisations that want to improve IDMP data management without replacing your current system.
Discover Our IDMP Resources

IDMP Timelines
[Downloadable PDF timeline]

IDMP Starter Pack
[Practical Guide]

Master Data Management
Best Practices
[White Paper]
The Impact of Ennov RIM at Foghorn Therapeutics
Efficient regulatory information management can be complex. Your software shouldn’t be.

How to Build a Strong Foundation for Commercial Growth
Learn how unifying Quality, Regulatory, and TMF archive management into one system eliminated ongoing CRO fees and helped teams move faster without adding headcount.



Aguettant Case Study
“It’s really nice to work with a software provider that shows a great deal of innovative spirit, has great ambition and is proactive. Above all, I really appreciate Ennov placing particular emphasis on the customer’s user experience.”
Cyrille Jeune,
Regulatory Affairs Systems Manager

Septodont Case Study
“With more than 1500 MAAs in 150 countries, we face a real productivity challenge. With Ennov, we have been able to issue 400 dossier in just 18 months. For the first time, our users are experiencing tremendous time savings when locating documents.”
Aurélie Becquet,
Regulatory Affairs
IDMP Webinar
The IDMP Submission Process
Create an IDMP submission in a few guided steps: select scope, generate the message, review changes, validate, and submit.
World-Class Regulatory Content and Information Management
A Regulatory suite with the power and flexibility to support the entire regulatory product lifecycle from the early planning of registration targets through to product retirement.
It is an invaluable solution for regulatory activity planning, product registration management, dossier creation, dossier management and more.
Why Choose Ennov
Over 500,000 users trust Ennov
Over 500,000 users trust Ennov
- Over 25 years of experience providing software solutions for Life Sciences and 450+ Life Science customers, with many more in other industries.
- Modern architecture and interface 100% web-based. Highly scalable. User-centric design.
- Our commitment to your success Very high customer satisfaction, 98.5% of projects delivered on time and within budget.
Providing you freedom of choice
Providing you freedom of choice
- Available as cloud-based or on-premises deployment, you can switch between deployment options at any time.
- System configuration and management require no IT skills, making you fully autonomous
- Improved security and optimized performance. Data can be hosted locally for total flexibility. Single-tenancy minimizes business interruptions.
Cloud-based or On Premises
Multi-Platform
ISO 9001 & 27001 Certified
How can we help you?
Fill out the form and we'll be in touch.



