Regulatory Publishing Software for Submissions & eCTD Publishing
Designed for Regulatory Operations and publishing teams, Ennov Dossier helps you build, validate, and publish regulatory submissions with compliant structure, hyperlinks, and traceable control from draft to archive.
- Create, manage, and publish regulatory submissions in a structured workflow
- Support eCTD publishing and other submission formats, including eCTD 4.0, NeeS, and ACTD
- Generate compliant hyperlinks and bookmarks to improve navigation and review
- Validate submission packages before delivery to reduce rework and delays
- Maintain a consistent archive with controlled versions and audit-ready history
The Submission Publishing Challenge
Global regulatory submissions require tight coordination across authoring, review, approval, publishing, and delivery, often across multiple teams and locations. When documents and submission components live in file shares and email threads, teams lose time to version checks, manual assembly, and last-minute fixes, increasing the risk of delays and rework.
Ennov Dossier, our regulatory publishing software helps address this by bringing submission publishing into a controlled workflow, with structured assembly, compliant hyperlinking and bookmarking, and validation checks before release. The goal is not only to publish faster, but to publish with more consistency, traceability, and fewer avoidable issues across submissions and lifecycle updates.
Efficient and Easy to Use
Regulatory Publishing Software
Ennov Dossier is a powerful regulatory publishing software that helps teams build, manage, publish, validate, and archive regulatory submissions using controlled content in Ennov Regulatory Documents. This reduces fragmented steps like locating files across shared drives, copying versions, and manually uploading documents for submission assembly, helping teams publish with more consistency and less rework.
Publishers can assemble submissions using a simple drag-and-drop interface to link documents into the correct structure quickly. Built-in validation, plus compliant hyperlinking and bookmarking, helps teams catch issues earlier and produce submission-ready outputs with confidence.
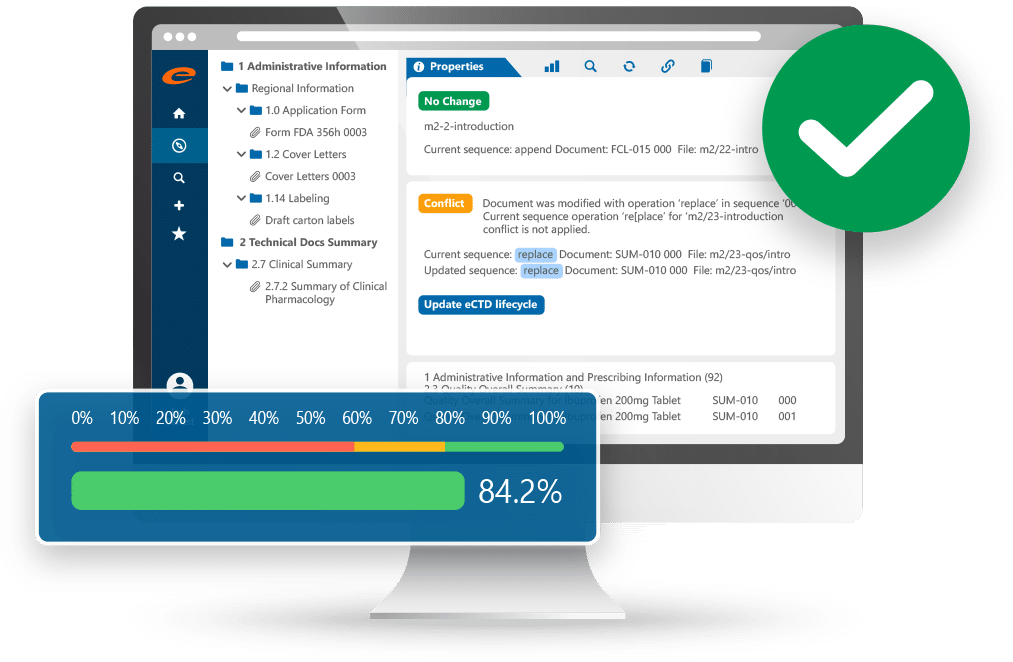
Improve Productivity with our Ennov's Integrated eDMS
When combined with Ennov’s powerful regulatory document management system, Ennov Dossier brings regulatory publishing software and EDMS capabilities together to support controlled dossier management from source content through published submissions. Teams can manage regulatory dossiers with structured metadata, traceability, and consistent control across documents, versions, and approvals.
Automated dossier lifecycles, workflows, and notifications reduce manual handoffs and help keep publishing work moving on schedule. Metadata-based navigation also improves productivity by helping users find dossiers by key properties, such as product, market, or submission type, rather than relying on folder structures and shared drives.
Compliant Submissions Every Time, With Ennov's Robust Regulatory Publishing Software
Ennov Dossier supports regulatory submission publishing with configurable submission assembly templates for regions that accept eCTD submissions, as well as non-eCTD formats. Templates can be tailored to client requirements and updated as regulatory guidance evolves.
To support efficient review, Ennov Dossier can generate tables of contents, hyperlinks, bookmarks, and other navigation aids. During publishing, the submission package is assembled with the required components, including ICH and regional XML files, correctly named leaf files, and appropriate folder structures, helping teams produce submission-ready outputs with greater consistency.
- CTD, eCTD, NeeS, VNeeS and eCopy support
- Dossier life cycle management
- eCTD sequence and metadata management
- Robust hyperlinking and bookmarking
- Integrated eCTD validator
- Built-in submission assembly templates
- Full text and metadata based searching
- Intrinsically connected with Ennov Regulatory Documents
- Intuitive drag-and-drop user interface
- Compatible with any WebDAV compliant repository
- Automatic compliant PDF rendering
- 100% web-based
Regulatory Publishing Software FAQs
What is regulatory publishing software?
Regulatory publishing software helps regulatory teams assemble, validate, and publish regulatory submissions in a compliant structure. It supports submission publishing workflows by organizing content into the right hierarchy, generating required components, and maintaining traceability from draft assembly through published output and archive.
What is submission publishing in Regulatory Operations?
Submission publishing is the process of assembling approved content into a submission-ready package for health authorities, following required structures, naming conventions, and technical rules. Publishing typically includes building the submission table of contents, creating hyperlinks and bookmarks, running validation checks, and producing the final output for delivery and lifecycle updates.
What is eCTD publishing software used for?
eCTD publishing software is used to publish electronic Common Technical Document submissions in the format required by agencies. It supports structured submission assembly, technical validation, and creation of required components such as XML files, correctly named leaf files, and folder structures, helping reduce rework before submission.
What formats does Ennov Dossier support for regulatory submissions?
Ennov Dossier supports multiple regulatory submission formats, including eCTD (3.2.2) and eCTD 4.0, as well as NeeS and ACTD. Submission assembly templates can be configured to align with regional requirements and updated as guidance changes.
What is the difference between dossier management and regulatory publishing software?
Dossier management focuses on organizing and controlling the underlying content and records over time. Regulatory publishing software focuses on turning approved content into a compliant submission package, including assembly, validation, hyperlinking, bookmarking, and output creation. In practice, publishing is most efficient when publishing workflows stay connected to controlled source content.
How does regulatory publishing software reduce rework and delays?
Publishing delays often come from fragmented handoffs, version confusion, manual assembly, and late discovery of technical errors. Regulatory submission publishing software reduces rework by using controlled content, structured templates, and validation checks during assembly, helping teams catch issues earlier and publish with greater consistency.
What publishing features support submission review and navigation?
For reviewers, navigation matters. Ennov Dossier supports common publishing elements that improve dossier usability, including tables of contents, hyperlinks, and bookmarks, helping reviewers move through content efficiently and reducing time spent manually creating navigation aids.
What should I look for when evaluating regulatory publishing software?
Look for support for your required formats (eCTD 3.2.2, eCTD 4.0, NeeS, ACTD), configurable templates, reliable validation, and the ability to generate required submission components. Also evaluate how well the software supports publishing workflows, traceability, and archiving, and whether it stays connected to controlled source documents to reduce duplicate work.
How does Ennov Dossier support archiving and lifecycle updates?
Regulatory publishing doesn’t end at the first submission. Ennov Dossier helps teams archive published submissions with consistent structure and controlled versions, supporting future lifecycle activities such as variations, responses, and ongoing submission updates.
How does Ennov Dossier work with Ennov Doc and DocShifter?
Ennov Dossier can be used alongside Ennov Doc to keep submission publishing connected to controlled source content and document governance. If DocShifter is part of your process, it can support document preparation and consistency checks before publishing, helping teams reduce manual formatting work and improve submission readiness.
Discover Our Submission Publishing Resources

eCTD 4.0 Pilots: Cleared for Takeoff!
[Read the Article]

eCTD Content Hub
[Practical Guide]

eCTD 4.0: What’s Changing and How to Be Ready
[White Paper]

eCTD 4.0 Pilot Readiness Checklist
[Blog Article]

Ennov Dossier Product Brief
[Webinar]

Which regulatory updates should you pay attention to?
[Product Demo]
The Impact of Ennov RIM at Foghorn Therapeutics
Efficient regulatory information management can be complex. Your software shouldn’t be.

How to Build a Strong Foundation for Commercial Growth
Learn how unifying Quality, Regulatory, and TMF archive management into one system eliminated ongoing CRO fees and helped teams move faster without adding headcount.



Aguettant Case Study
“It’s really nice to work with a software provider that shows a great deal of innovative spirit, has great ambition and is proactive. Above all, I really appreciate Ennov placing particular emphasis on the customer’s user experience.”
Cyrille Jeune,
Regulatory Affairs Systems Manager

Septodont Case Study
“With more than 1500 MAAs in 150 countries, we face a real productivity challenge. With Ennov, we have been able to issue 400 dossier in just 18 months. For the first time, our users are experiencing tremendous time savings when locating documents.”
Aurélie Becquet,
Regulatory Affairs
World-Class Regulatory Content and Information Management
A Regulatory suite with the power and flexibility to support the entire regulatory product lifecycle from the early planning of registration targets through to product retirement.
It is an invaluable solution for regulatory activity planning, product registration management, dossier creation, dossier management and more.
Why Choose Ennov
Over 500,000 users trust Ennov
- Over 25 years of experience providing software solutions for Life Sciences and 450+ Life Science customers, with many more in other industries.
- Modern architecture and interface 100% web-based. Highly scalable. User-centric design.
- Our commitment to your success Very high customer satisfaction, 98.5% of projects delivered on time and within budget.
Providing you freedom of choice
- Available as cloud-based or on-premises deployment, you can switch between deployment options at any time.
- System configuration and management require no IT skills, making you fully autonomous
- Improved security and optimized performance. Data can be hosted locally for total flexibility. Single-tenancy minimizes business interruptions.
How can we help you?
Fill out the form and we'll be in touch.
