Clinical Data Management Software for Clinical Trial Data Capture and Control
Designed for clinical operations and data management teams, Ennov’s clinical data management software supports EDC, RTSM, and ePRO workflows to capture and validate clinical trial data with consistent governance and traceability.
- Capture and manage study data in a structured CDMS foundation
- Support RTSM workflows to keep randomization and supply aligned to the study
- Collect patient-reported data through integrated ePRO
- Improve data quality with validations, edit checks, and audit-ready traceability
- Easily import data, clean exports, and trace data reliablely
- Gain clearer oversight with reliable exports, reporting, and study visibility
Benefits
- One unified platform
Manage clinical data, documents, workflows, and oversight within a single, integrated suite. Simplify operations and eliminate silos across systems and teams. - Fewer manual tasks, greater productivity
Automate repetitive processes like data validation, site payments, and monitoring visit reports, to free up time and reduce risk of error.
- Designed for teams of any size, anywhere
Whether you’re running one study or dozens, Ennov supports collaboration across regions, teams, and partners, with intuitive tools and multilingual support built in. - Real-time visibility, better decisions
Access dashboards and analytics that provide actionable insights into trial progress, site activity, and compliance risks, before they become costly problems.
The clinical data capture and data quality challenge
Clinical trial data capture is only valuable when it is consistent, reviewable, and ready for downstream analysis. When data collection and management rely on manual handoffs, disconnected tools, or inconsistent definitions across sites, teams lose time to reconciliation, query cycles, and late data cleaning, and oversight becomes harder as study complexity grows.
Clinical data management software (CDMS) helps address this by providing a structured foundation for capturing, validating, and tracking protocol-specific data throughout a study. Within that foundation, electronic data capture (EDC) supports efficient data entry and review through controlled forms and edit checks, faster access to current study data, stronger security, and clearer visibility for sponsors, CROs, and sites. Selecting the right approach matters, because flexibility, usability, and compliance expectations vary across trial designs and operating models.
A strong clinical data management foundation depends on reliable electronic data capture, consistent validation, and clear oversight across sites. Ennov EDC is designed to support that from study build through data capture and review.
A Complete Solution for Clinical Trial Data Capture and Management
Ennov EDC is part of Ennov’s clinical data management software offering, supporting electronic data capture for protocol-specific study data with controlled forms and built-in checks to reduce missing or inconsistent entries. It helps clinical teams design studies, deploy them across sites, and capture subject data in a structured, reviewable way.
Designed for multi-center trials, Ennov EDC supports flexible study build and scalable deployment across different trial types and phases, including early-phase and late-phase studies, post-marketing programs, and observational designs. By improving data consistency and visibility across sites and stakeholders, Ennov EDC helps teams reduce rework, support faster review cycles, and improve coordination across clinical operations.

A connected approach to clinical data management
Clinical trial teams depend on accurate, consistent data across many activities, from data capture and randomization through trial operations and trial documentation. When information is spread across disconnected systems, teams lose time to manual reconciliation, duplicate entry, and inconsistent definitions that slow oversight and reporting.
Ennov’s clinical data management software is designed to reduce that friction by connecting clinical data and documents on one platform. A shared foundation helps teams maintain consistent data, improve traceability, and support cross-functional visibility across clinical workflows.
If your organization already has a master data management approach in place, Ennov can align to it to exchange and use data consistently. If not, Ennov still supports structured data management through configurable data models and controlled processes that help teams maintain consistency over time.
- Quick and easy eCRF design
- Support for all data field formats
- Computed data fields and interval calculations
- Defined data field groups
- Dynamic data field activation
- Support for optional eCRF pages
- Configurable library of allowable values
- Online or offline patient data entry
- Attach photos to eCRF using iPad camera
- Streamlined clinical data capture
- Requires no IT or programming skills
- Increased data visibility and security
- Medical coding for MedDRA and WHO Drug
- CDISC, CDASH and SDTM compliant
- Full web interface
- 21 CFR Part 11 compliant
- Available in the App Store
- Compatible with existing Ennov Clinical studies
The Randomization and Trial Supply Management Challenge
Randomization is essential in many clinical trials, but execution becomes harder as studies add more sites, cohorts, and protocol changes. Teams need to assign subjects consistently, maintain allocation integrity, and keep a clear, traceable history of assignments across the study lifecycle.
Trial supply management adds another layer of operational complexity. Forecasting, packaging, labeling, distribution, and resupply must stay aligned with enrollment and site activity. When supply planning is managed across spreadsheets or disconnected tools, it becomes easier to create delays, shortages, or mismatches that can disrupt study timelines.
Ennov RTSM: Randomization and trial supply, in one workflow
Ennov RTSM is part of Ennov’s clinical data management software offering and supports both randomization and trial supply management in a single, controlled workflow. Through an integrated IWRS, investigators and site teams can complete study-specific activities through a web-based interface with role-based access and traceable actions.
Ennov RTSM supports complex randomization schemes, including stratification and minimization, and can be configured for common trial designs such as blinded and multi-arm studies. When used alongside Ennov EDC, randomization and key study data remain aligned, helping teams reduce manual handoffs and maintain consistent execution across sites.

For trial supply, Ennov RTSM supports management of IMP (investigational medicinal product) from initial shipment through dispensing and resupply. Stock visibility and notifications help site teams monitor inventory status and support timely replenishment, reducing the risk of avoidable supply disruption during study conduct.
Flexible and Configurable RTSM Workflows
Randomization and clinical data capture work best when they stay connected. That’s why Ennov RTSM can connect directly with Ennov EDC, helping investigators randomize eligible subjects as part of the same study workflow. Investigators can add a subject, confirm eligibility and stratification parameters, and request randomization through a controlled process, supporting consistent execution across sites.
Ennov RTSM supports configurable randomization setup, including stratification and minimization, plus options to manage blinding rules and randomization scenarios based on study design. Data managers can define how randomization is requested, which parameters are used, and how treatment assignment is managed across arms and strata, while maintaining traceability of each allocation decision.
When needed, randomization outcomes can be made available within the eCRF, such as treatment arm or required references for dispensing, helping sites follow the correct procedures without manual handoffs.
Core Capabilities
- Randomization control with consistency checks and email alerts
- Complete traceability of sudies, patients and status
- Integrated IWRS functionality
- Flexible randomization scenarios
- Support for any study type
- Clinical supply management
- Treatment allocation
- Blind management
Key Features
- Unlimited number of strata and treatment options
- Display randomization results in eCRF
- User friendly and easy to use
- Integrated with Ennov EDC
- Global follow-up with email alerts
- Flexible and configurable
- 100% web-based interface
- 21 CFR Part 11 compliant
The Patient-reported data collection challenge
Collecting patient-reported outcomes through paper diaries and questionnaires creates avoidable friction for both patients and study teams. Manual entry increases the risk of incomplete responses, inconsistent timing, and delayed visibility into what is happening in the field. It also introduces additional effort for sites and data managers, especially when paper responses must be transcribed into clinical systems and then cleaned and verified.
ePRO helps address these challenges by digitizing patient questionnaires and diaries in a controlled workflow. Electronic capture can support better timeliness, clearer traceability, and faster review of patient-reported data, helping teams reduce delays related to missing data and late corrections while improving study oversight.
Ennov ePRO, a Digital Alternative to Paper Diaries
Ennov ePRO is part of Ennov’s clinical data management software offering and supports electronic patient-reported data capture in a structured, traceable way. Patients can complete diaries and questionnaires through a user-friendly web application, while built-in checks help reduce incomplete or inconsistent entries. Patient-reported data becomes available to investigators and site teams to support monitoring of completion status and follow-up as needed.
Ennov ePRO supports common ePRO use cases such as patient self-assessments and quality-of-life questionnaires, with clear navigation designed to reduce user friction. It also supports Visual Analog Scale (VAS) questionnaires for data collected on a graduated scale. Notifications can be configured to prompt patients when new questionnaires are available, helping teams improve timeliness and reduce missed entries.
Ennov ePRO can also support veterinary studies where owners enter observations such as behavior or feeding information.
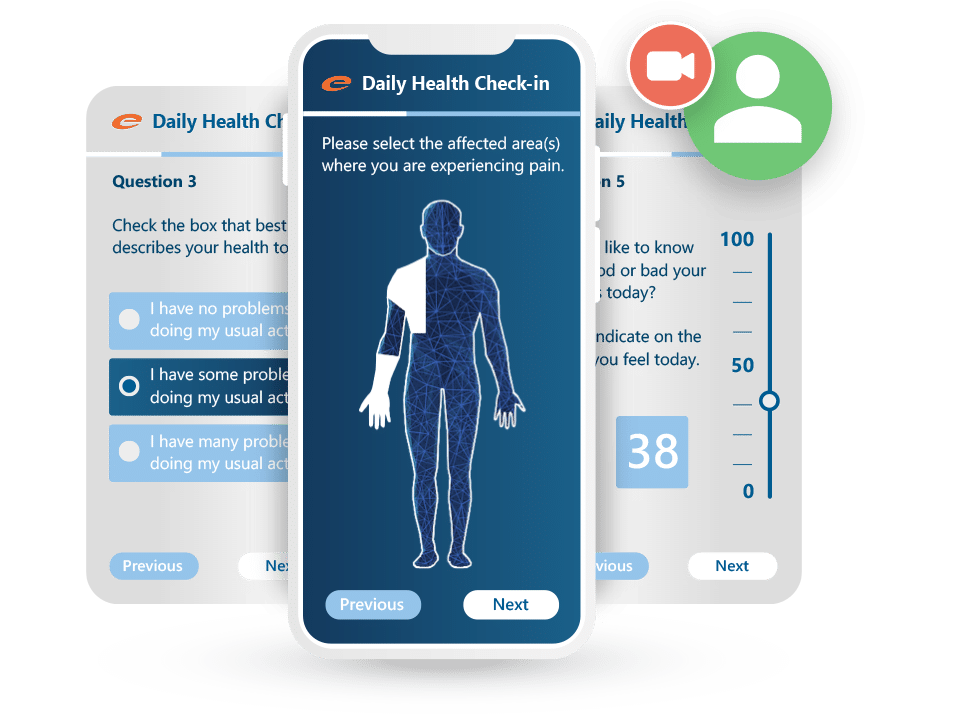
Safe, Secure, and Compliant ePRO Software
Ennov ePRO is designed to support secure capture of patient-reported data, with controls that help protect patient privacy and confidentiality. ePRO data can be encrypted and managed with role-based access, helping teams limit access to authorized users and maintain traceable handling of study data.
Ennov ePRO also supports compliance needs for regulated clinical research, including features commonly required for 21 CFR Part 11, such as audit trails and electronic signatures, depending on your configuration and validation approach.
Core Capabilities
- Intuitive and easy to use interface
- Configurable questionnaires
- Visual Analog Scale for self assessments
- Secure and encrypted
- Real time access to patient data
- Automated notifications
Key Features
- Fully integrated with Ennov EDC
- Saves time and money
- Higher patient compliance levels
- Increased data quality
- 100% web-based
- 21 CFR Part 11 compliant

How to Build a Strong Foundation for Commercial Growth
Learn how unifying Quality, Regulatory, and TMF archive management into one system eliminated ongoing CRO fees and helped teams move faster without adding headcount.




When Academic Excellence Meets Regulatory Reality




Building a Complete Research Operations Platform
How an academic research group replaced fragmented, outdated tools with a self-configured, unified platform covering study management, quality, eTMF, and analytics.



Clinical Data Management Software FAQs
What is clinical data management software?
Clinical data management software helps clinical teams capture, validate, and manage clinical trial data in a structured, traceable way. It supports consistent data collection across sites, improves oversight during study conduct, and helps teams maintain data quality throughout the trial lifecycle.
What is clinical data management software used for?
Clinical data management software is used to design study data capture, apply edit checks and validation, manage changes over time, and support clean exports and reporting. It helps reduce manual reconciliation and improves visibility into study progress and data completeness.
What should I look for in clinical data management software for regulated trials?
Look for strong data validation, audit-ready traceability, role-based access, and the ability to configure workflows to match your study design and SOPs. It should also support consistent oversight across studies and sites and scale across different trial types and operating models.
How does clinical data management software support data quality and traceability?
Clinical data management software supports data quality through controlled forms, validations, and consistent rules applied during data capture and review. Traceability comes from maintaining an auditable history of updates, approvals, and changes so teams can understand what changed, when, and why.
Who typically uses clinical data management software?
Common users include data managers, clinical trial managers, CRAs/monitors, biostatisticians, and site teams. Sponsors, CROs, and research sites rely on clinical data management software to coordinate data capture and maintain study oversight across geographies.
How does Ennov support EDC within its clinical data management software offering?
Ennov’s clinical data management software includes EDC capabilities for structured clinical trial data capture using configurable forms and built-in checks. This helps teams reduce missing or inconsistent entries and supports efficient study build and execution across sites.
How does RTSM fit into clinical data management software?
RTSM supports randomization and trial supply workflows that need to stay aligned with study execution and enrollment status. As part of a connected clinical setup, RTSM helps teams manage allocation rules and supply operations with traceable processes and clearer operational visibility.
What is ePRO software, and how does it support clinical trials?
ePRO software supports electronic capture of patient-reported outcomes such as diaries and questionnaires. It helps improve timeliness and completeness of patient-reported data and supports oversight by making completion status and responses available to study teams in a structured workflow.
Efficiently & Securely Capture & Manage Clinical Trial Information
The Ennov Clinical suite consists of Clinical Data Management applications as well as Clinical Trial Management applications that are available for deployment in the cloud or on premises.
Why Choose Ennov
Over 500,000 users trust Ennov
- Over 25 years of experience providing software solutions for Life Sciences and 450+ Life Science customers, with many more in other industries.
- Modern architecture and interface 100% web-based. Highly scalable. User-centric design.
- Our commitment to your success Very high customer satisfaction, 98.5% of projects delivered on time and within budget.
Providing you freedom of choice
- Available as cloud-based or on-premises deployment, you can switch between deployment options at any time.
- System configuration and management require no IT skills, making you fully autonomous
- Improved security and optimized performance. Data can be hosted locally for total flexibility. Single-tenancy minimizes business interruptions.
How can we help you?
Fill out the form and we'll be in touch.
