Ennov CTMS
Clinical Trial Management Software
Designed for clinical teams of all sizes, Ennov’s CTMS software centralizes operational data to ease the planning, management and reporting of clinical trials.
- Centralize clinical trial information and records in one CTMS software
- Strengthen compliance with controlled workflows, traceability, and audit-ready documentation
- Simplify planning, management and reporting of clinical trials
- Reduce manual effort with automated assignments, approvals, and reminders
- Fully integrated with Ennov clinical data management applications for faster study startup & increased data quality
- Understand your data thanks to pre-configured views, reports and dashboards
What challenges does CTMS software solve?
Clinical trials often involve dozens or even hundreds of investigator sites across multiple regions. When operational data is spread across spreadsheets, emails, and disconnected systems, teams lose visibility into site performance, enrollment status, monitoring activity, and upcoming deadlines.
CTMS software (Clinical Trial Management System software) helps solve this by centralizing operational trial data in one system of record. It supports site selection and activation, recruitment and screening tracking, visit planning and scheduling, financial tracking, and portfolio-level reporting, so clinical operations teams can manage studies more efficiently and make decisions based on accurate, up-to-date metrics.
Ennov CTMS: A single authoritative source for clinical trials
Ennov CTMS facilitates the end-to-end management of clinical trials. The software allows sponsors to be more efficient, to make better decisions, to ensure compliance, to select investigators well, to monitor patient recruitment and to manage finances. Ennov CTMS includes comprehensive directories to manage site personnel (investigators, sub-investigators, study coordinators, pharmacists) and organizations (hospitals, competent authorities, CROs, suppliers).
Ennov CTMS provides functions for tracking patients, patient visits, EDC data, activities, queries, deviations, adverse events, drug supply, monitoring visits and finances.
A full complement of reports and dashboards provide total visibility into the status of each study, country, site and investigator to provide the information necessary to make the best decisions possible.
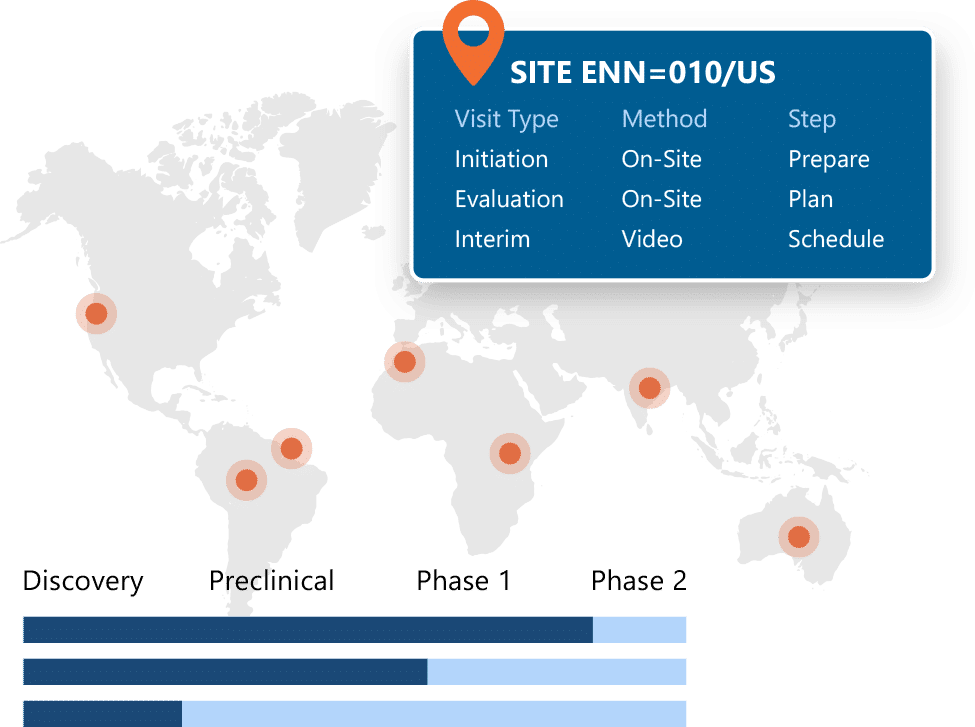
Centralized and Globally Accessible CTMS Software
Ennov CTMS is built on our Unified Compliance Platform, including Ennov Workflow for business process management, Ennov Doc for document management, and Ennov Analytics for reporting and data visualization, to provide a centralized CTMS software foundation for clinical operations. It supports secure access for internal teams and external partners through configurable role-based permissions, enabling collaboration while maintaining control over clinical trial information.
Ennov CTMS also integrates with Ennov Clinical Data Management applications. Study information from Ennov EDC can be imported into Ennov CTMS to support faster study startup and reduce duplicate data entry. By consolidating operational trial data in a single repository, trial managers gain clearer visibility across studies, improve data consistency, and make more informed decisions based on up-to-date information.
Advanced Analytics for Risk-Based Management of Clinical Trials
Risk-based approaches depend on timely operational data to identify issues early and keep studies on track. Ennov CTMS includes CTMS dashboards that bring key trial information together to show status, progress, risks, and outliers across your portfolio.
Dashboards can provide visibility into recruitment and enrollment, site and subject status, protocol deviations and CAPAs, health authority status, adverse events, and query trends, depending on your configuration and data sources. Many dashboards reflect recommendations from industry initiatives such as TransCelerate. Users can filter views by study, country, and investigational site, then export results to support ongoing reporting and team reviews.
Core Capabilities
- Comprehensive organization and personnel directories
- Patient recruitment tracking
- Query and deviation tracking
- Adverse event tracking
- Drug supply management
- Financial and budget trackling
- Robust monitoring capabilities
Key Features
- Centralized database
- Integrated workflow
- Comprehensive and configurable data model
- Intuitive user interface
- Intergrated with Ennov CDMS
- 100% web-based
- 21 CFR Part 11 compliant
CTMS Software FAQs
What is CTMS (clinical trial management) software?
CTMS software (Clinical Trial Management System software) is clinical trial software used to manage operational trial activities and data across studies and sites. It centralizes information such as study milestones, site status, monitoring activity, recruitment progress, documents, tasks, and reporting so teams can coordinate work and maintain visibility throughout the clinical trial lifecycle.
What is CTMS (clinical trial management software) used for?
Clinical trial management software is used to plan, track, and report on trial execution. Common use cases include site selection and activation tracking, visit and monitoring planning, recruitment and enrollment oversight, issue and action tracking, document coordination, and operational reporting, including CTMS dashboards that highlight status, risks, and outliers across studies.
What processes does Ennov CTMS support out of the box?
Ennov CTMS supports core clinical operations workflows out of the box through configurable processes and structured data capture. Typical out-of-the-box CTMS workflows include study setup and planning, site onboarding and status tracking, activity and milestone tracking, monitoring and visit scheduling support, task routing and approvals, and operational reporting through dashboards. Organizations can then configure roles, fields, and workflows to align with their SOPs and operating model.
What is the Ennov CTMS core model Model, and what are the benefits?
The Ennov CTMS core model is a preconfigured foundation that provides a starting structure for CTMS data, workflows, roles, and reporting patterns. The benefit is faster implementation, less build-from-scratch effort, and more consistent rollout across teams and studies. It also supports better data consistency and governance over time, which improves reporting reliability and day-to-day trial oversight.
What is the difference between a CTMS system and CTMS system software?
A CTMS system refers to the full way an organization runs clinical trial management, including people, SOPs, governance, and processes. CTMS system software is the technology that supports that system by centralizing operational data, enforcing workflows, controlling access, and enabling dashboards and reporting. In practice, the software helps teams execute the CTMS system consistently across studies and sites.
What should I look for in CTMS compliance software for regulated life sciences?
For regulated environments, CTMS compliance software should support controlled workflows, role-based access, traceability of updates and decisions, and reliable reporting. Look for the ability to standardize processes across studies while still allowing configuration to match SOPs, and make sure the platform can support inspection readiness through structured records, audit-friendly history, and consistent documentation practices.
Who are the main users of Ennov's CTMS (clinical trial management software)?
Ennov’s CTMS software is used by clinical operations and study management teams across different trial stakeholders, including Sponsors, CROs, and research sites. Typical users include study managers, clinical trial managers, CRAs/monitors, site coordinators, and operational leadership who rely on CTMS dashboards and reporting to track progress, workload, and risks across studies and sites.
How an Academic Research Center Delivered Pharma-Grade Trial Performance



Efficiently & Securely Capture & Manage Clinical Trial Information
The Ennov Clinical suite consists of Clinical Data Management applications as well as Clinical Trial Management applications that are available for deployment in the cloud or on premises.
Why Choose Ennov
Over 500,000 users trust Ennov
- Over 25 years of experience providing software solutions for Life Sciences and 450+ Life Science customers, with many more in other industries.
- Modern architecture and interface 100% web-based. Highly scalable. User-centric design.
- Our commitment to your success Very high customer satisfaction, 98.5% of projects delivered on time and within budget.
Providing you freedom of choice
- Available as cloud-based or on-premises deployment, you can switch between deployment options at any time.
- System configuration and management require no IT skills, making you fully autonomous
- Improved security and optimized performance. Data can be hosted locally for total flexibility. Single-tenancy minimizes business interruptions.
Cloud-based or On Premises
Multi-Platform
ISO 9001 & 27001 Certified
How can we help you?
Fill out the form and we'll be in touch.
