Ennov Pharmacovigilance Suite
An AI-powered, end-to-end solution for collecting, reporting and analyzing human
and vet PV data
Used by pharma companies, CROs, and health authorities around the world, Ennov’s AI-powered Pharmacovigilance suite brings everything together (from case intake to signal detection) in one unified system. You can collect, manage, assess, and report human and veterinary adverse events in one seamless workflow — with built-in tools for advanced analysis and smarter decisions.
- A single authoritative source
Manage and track all pharmacovigilance data within a single, unified database to streamline PV operations and ensure regulatory compliance. - Improved performance
Eliminate manual, paper-based processes and record keeping. Automate PV case intake and reporting to achieve productivity gains.
- Ensure compliance
Gain total confidence that all aspects of achieving and maintaining regulatory compliance, including electronic reporting, will be met. - Increased visibility
Get a real-time overview of your safety database with dynamic metrics and dashboards.
“We were happy to find the support and respect Ennov demonstrated, which we felt was quite rare in a shared business partnership.”
Phil T.,
Business Development Manager, Pure Drug Safety
Doc for Pharmacovigilance
Centralize your PV documentation in one compliant system
Ennov Doc for PV centralizes documents and the PSMF, while supporting agreement management in a structured repository built for compliance.
Why teams choose Ennov Doc for Pharmacovigilance:
- Stay inspection-ready with one system for PV documents, PSMF, and agreements
- Reduce compliance risk with controlled lifecycles, ownership, and audit trails
- Keep the PSMF audit-ready with a structured binder aligned to global requirements
- Centralize submissions and reports to support global PV reporting needs
- Manage risk documentation consistently across global and local teams
- Meet 21 CFR Part 11 with compliant controls for regulated PV work

Case Intake & Management
Capture confidently. Configurable workflows. Stay compliant.
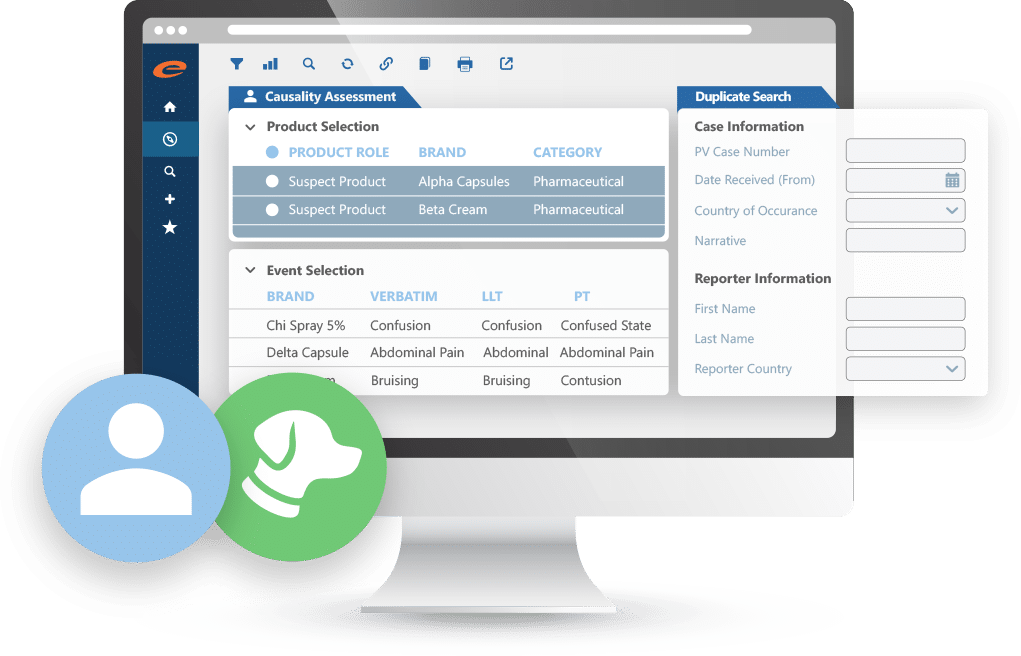
Efficiently manage human and animal pharmacovigilance case data from any source (clinical trial or post-marketing) with Ennov’s AI-powered, flexible, compliant solution. Import reports in various formats or enter them directly using fast, intuitive templates. Configurable workflows keep cases on track from intake through coding, assessment, and reporting.
Why teams choose Ennov for case intake and management:
- Accurately capture and manage both human and veterinary PV cases
- Import adverse event data or enter directly using flexible templates
- Fully configurable workflows to support end-to-end case processing
- Seamlessly integrates into existing pharmacovigilance operations
- Trusted by pharma, CROs, service providers, and regulators
- Scalable, compliant, and designed to fit your business processes
- AI-powered data extraction and triage to streamline intake and prioritize high-risk cases
Reporting & Data Analysis
Compliant by default. Fast by design. Global by nature.
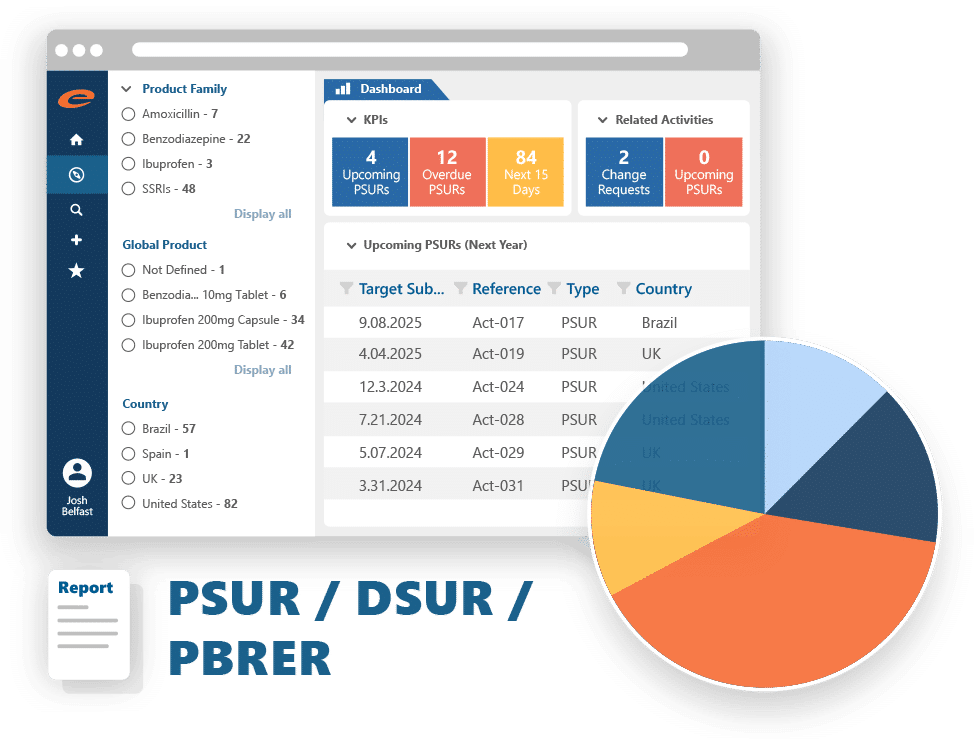
A compliant, easy-to-use pharmacovigilance tool giving you flexibility, automation, and confidence — without the typical complexity of reporting.
Why teams choose Ennov for compliant reporting and analysis:
- Generate data extracts, paper reports, and XML submissions with ease
- Analyze safety data using pre-supplied or custom-built queries
- Seamlessly submit to global health authorities including FDA and EMA
- Integrated E2B-compliant gateway streamlines electronic submissions
- Supports all standard single case and aggregate reports
- Offers both manual and automated generation and submission options
Signal Detection & Management
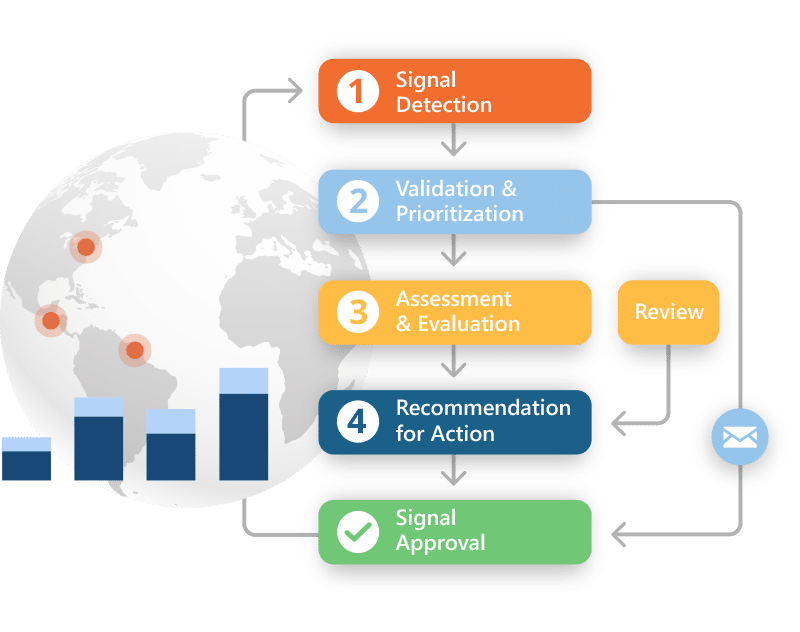
Spot the signal. See the trend. Stay ahead.
Intuitive analysis helps you identify trends, track signal activity over time, and stay aligned with internal risk processes. All without complex setup or customization.
Why teams choose Ennov for Signal Detection and Management:
- Out-of-the-box signal detection and data mining for business users
- Access to advanced statistics, data cubing, and visualizations
- Calculate PRR, ROR, and MGPS to assess safety signals
- Apply smart stratification to surface threshold-based trends
- Searchable repository to track and review signal evolution
- Configurable workflows aligned with internal risk processes
Pharmacovigilance Software FAQs
What is pharmacovigilance software?
Pharmacovigilance software is a drug safety system used to manage safety data and PV processes across a product’s lifecycle. It helps teams capture and process cases, maintain compliant records, support reporting timelines, and monitor safety trends with consistent workflows and traceability.
What is pharmacovigilance software used for?
Pharmacovigilance software is used to support end-to-end PV operations, including case intake and triage, coding, medical review, submissions and reporting, signal detection and management, and ongoing safety oversight. It helps PV teams reduce manual work, maintain data quality, and stay inspection-ready across global operations.
What is ICSR case management, and how does pharmacovigilance software support it?
ICSR (Individual Case Safety Report) case management covers the process of capturing adverse event information, assessing seriousness and expectedness, coding relevant terms, performing medical review, and maintaining a complete case history. Pharmacovigilance software supports this by standardizing intake, routing tasks for review and QC, preserving audit trails, and enabling efficient follow-up and reporting.
What is case intake in pharmacovigilance?
Case intake is the process of receiving potential safety information from sources such as spontaneous reports, literature, clinical programs, partners, or medical information channels. Pharmacovigilance software helps by structuring intake, capturing the required minimum information, supporting deduplication and triage, and routing cases into the right workflow for assessment.
What is expedited reporting in pharmacovigilance?
Expedited reporting refers to time-sensitive reporting requirements for specific case types, typically based on seriousness, expectedness, and timelines defined by regulations and SOPs. Pharmacovigilance software helps teams manage expedited reporting by tracking deadlines, supporting workflows for review and QC, and maintaining consistent case documentation for inspection readiness.
What is signal detection in pharmacovigilance, and what role does software play?
Signal detection is the process of identifying potential new safety concerns by reviewing trends and patterns across safety data sources. Pharmacovigilance software supports signal detection by consolidating safety data, enabling structured review and documentation of signal evaluation steps, and supporting governance around decisions, actions, and follow-up.
What is a PSMF in pharmacovigilance, and how can software help?
A PSMF (Pharmacovigilance System Master File) describes how a company’s PV system is organized, including processes, responsibilities, and supporting documentation. Pharmacovigilance software can help by maintaining controlled, inspection-ready PV documentation, ensuring consistency across updates, and supporting traceability of approvals and revisions.
What is a QPPV, and why does a PV system matter?
The QPPV (Qualified Person Responsible for Pharmacovigilance) role is responsible for oversight of the PV system and ensuring it operates effectively. A PV system matters because it provides visibility, governance, and documentation needed to demonstrate oversight, compliance, and control across PV activities and safety documentation.
What is the difference between pharmacovigilance software and drug safety software?
In practice, the terms are often used interchangeably. “Drug safety software” usually refers to the technology supporting PV processes, while “pharmacovigilance software” may emphasize regulatory and lifecycle safety obligations. The important evaluation point is whether the platform supports your required PV workflows, documentation, and compliance needs across the lifecycle.
What pharmacovigilance processes does Ennov support?
Ennov supports the full pharmacovigilance lifecycle, including case intake and management, signal detection and management, risk management activities, regulatory reporting support, and PV quality and governance processes. Ennov also supports PV documentation management, helping teams keep PSMF, procedures, agreements, and safety documentation organized, controlled, and inspection-ready.
How does Ennov support pharmacovigilance documentation management?
In addition to pharmacovigilance workflows, Ennov includes Doc for PV to manage PV-related documentation such as PSMF content, procedures, safety agreements, reports, and supporting evidence. It helps teams maintain controlled documents with traceability, version control, and consistent access across PV stakeholders.
Does Ennov support pharmacovigilance for human and veterinary products?
Yes. Ennov’s pharmacovigilance software is designed to support PV operations for both human and veterinary products, with configurable workflows and documentation controls to align with your organization’s processes and regulatory obligations.
How can we help you?
Fill out the form and we'll be in touch.
