
Clinical trials are more complex than ever in 2026, generating massive volumes of structured and unstructured data. Sponsors and CROs must ensure that this data is accurate, compliant, and submission-ready.
That’s why understanding what is clinical data management and how modern platforms like Ennov support it is critical for successful clinical operations.
If you’re asking what is clinical data management, this guide explains the full process, technologies, challenges, and best practices shaping clinical trials in 2026.
What Is Clinical Data Management?
Clinical data management is the process of collecting, validating, cleaning, integrating, and managing clinical trial data to ensure it is accurate, complete, and compliant with global regulatory standards.
The primary objective of clinical data management is to transform raw clinical trial data into trusted, submission-ready evidence that supports:
- High-quality datasets
- Reliable statistical outcomes
- Regulatory compliance
- Faster clinical decision-making
Modern clinical data management platforms help organizations centralize data workflows, reduce errors, and maintain full traceability across clinical studies.
What Is Clinical Data Management and Why Does It Matter?
Clinical data management plays a critical role in ensuring clinical trial success.
Ensures Data Integrity
Accurate and consistent data is essential for evaluating treatment safety and efficacy.
Supports Regulatory Compliance
Health authorities require traceable and auditable datasets aligned with standards such as:
- ICH GCP
- FDA 21 CFR Part 11
- CDISC
Improves Trial Efficiency
Streamlined workflows reduce delays and accelerate time-to-market.
Enhances Patient Safety
Real-time access to clean data helps organizations identify adverse events and operational risks faster.
Platforms like Ennov help organizations improve clinical oversight through integrated and compliant clinical environments.
The Clinical Data Management Process Explained
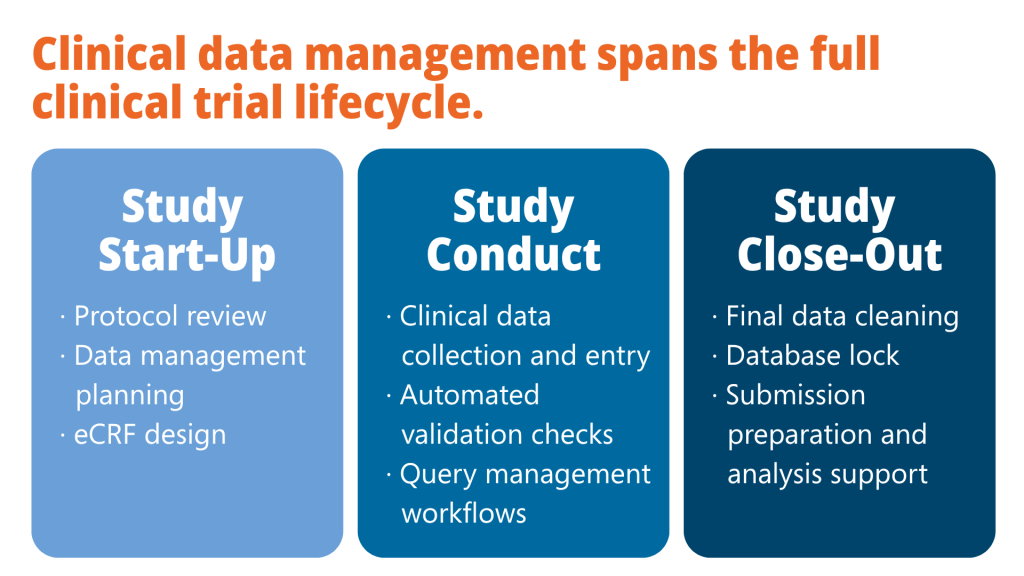
Modern clinical data management software unifies these activities into a controlled environment, reducing manual effort and improving operational visibility.
Key Components of Clinical Data Management
Data Collection
Clinical trial data is captured through:
- Electronic Data Capture (EDC) systems
- Lab integrations
- ePRO platforms
- Wearables and connected devices
Data Validation
Automated edit checks and validation rules ensure consistency and accuracy.
Data Cleaning
Ongoing query management and discrepancy resolution improve data readiness.
Data Integration
Clinical data management systems integrate data from multiple sources, including labs, imaging systems, and external vendors.
Database Lock
Finalized datasets are locked before statistical analysis and submission activities begin.
Ennov centralizes these components within a unified platform to improve traceability and reduce fragmentation.
Technologies Used in Clinical Data Management in 2026
Modern clinical data management relies heavily on advanced technologies.
Artificial Intelligence (AI)
AI helps detect anomalies, automate validation, and improve data cleaning workflows.
Cloud-Based Platforms
Cloud systems enable global collaboration and remote access to study data.
Decentralized Trial Support
Modern systems support hybrid and decentralized clinical trials through remote data capture capabilities.
Automation
Workflow automation reduces manual entry, accelerates query resolution, and improves operational efficiency.
Platforms like Ennov embed these technologies into unified clinical ecosystems to support scalable clinical operations.
How Ennov Supports Clinical Data Management
One of the biggest challenges in clinical data management is disconnected systems. Ennov addresses this challenge with an integrated clinical platform.

This unified approach eliminates silos and improves consistency across clinical operations.
Built-In Compliance
Ennov supports:
- GxP compliance
- Audit trails
- Data integrity requirements
- Inspection readiness
Configurable Workflows
Teams can adapt workflows to study-specific requirements without extensive customization.
Real-Time Visibility
Dashboards and reporting tools provide immediate insight into trial progress and data quality.
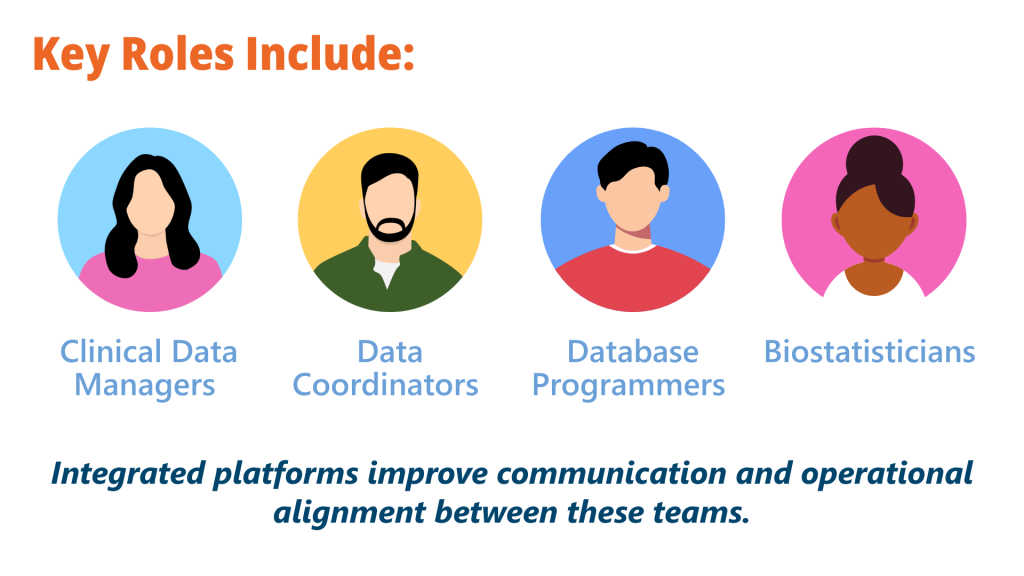
Roles in Clinical Data Management
Effective clinical data management requires collaboration across multiple specialized roles.
Challenges in Clinical Data Management in 2026
Organizations continue to face several operational and regulatory challenges.
Common Challenges
- Increasing data volume and complexity
- Managing multiple external data sources
- Maintaining real-time data quality
- Meeting evolving regulatory expectations
Unified clinical data management platforms help organizations centralize processes and standardize oversight.
Why Choose Ennov for Clinical Data Management?

This makes Ennov a strong option for organizations modernizing their clinical data management strategy.
The Future of Clinical Data Management
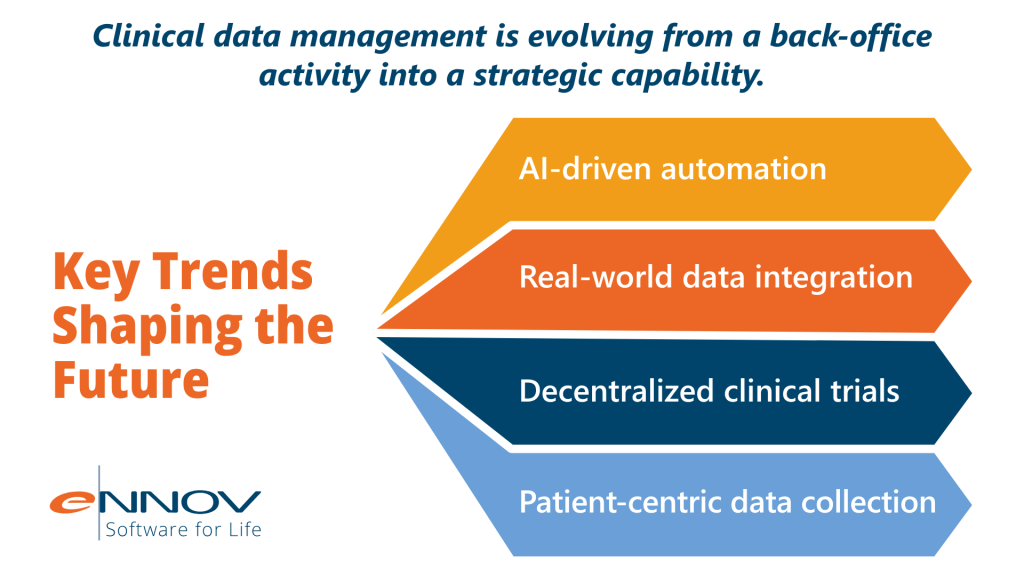
Modern platforms like Ennov help organizations manage increasing complexity while maintaining compliance and operational efficiency.
FAQs About Clinical Data Management
What is clinical data management used for?
Clinical data management is used to collect, validate, clean, and manage clinical trial data while ensuring accuracy, integrity, and regulatory compliance.
Why is clinical data management important?
Clinical data management improves data quality, supports regulatory submissions, enhances patient safety, and accelerates clinical trial timelines.
What software is used in clinical data management?
Organizations use clinical data management systems (CDMS), EDC platforms, CTMS software, and integrated clinical solutions like Ennov.
What is the difference between CDMS and EDC?
EDC systems focus on electronic data capture, while CDMS platforms manage the broader process of validation, cleaning, integration, traceability, and submission readiness.
What are the phases of clinical data management?
The main phases include study start-up, data collection, validation, query management, database lock, and submission preparation.
Conclusion
So, what is clinical data management?
Clinical data management is the foundation of reliable clinical research, ensuring every data point collected during a clinical trial is accurate, compliant, traceable, and actionable.
In 2026, successful clinical trials depend not only on collecting data, but on managing it intelligently across integrated systems.
With unified platforms like Ennov, organizations can streamline operations, improve data quality, maintain compliance, and accelerate the path to market.


