
If you’re evaluating a modern clinical data management system, here’s what to look for in 2026, especially with a focus on regulatory compliance, EDC/RIM integration, and audit trails.
Why a Clinical Data Management System Matters
A clinical data management system sits at the center of clinical operations and directly impacts:
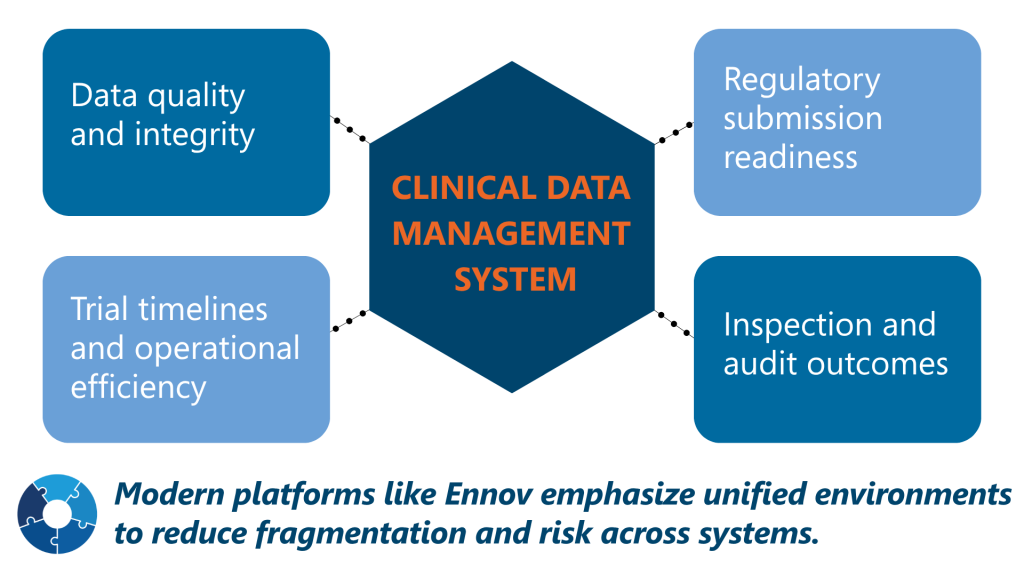
Clinical Data Management System Compliance Requirements
Regulatory compliance is one of the most critical factors when selecting a clinical data management system. Your platform must support global standards and maintain inspection readiness at all times.
Key Compliance Requirements
Look for systems aligned with:
- FDA 21 CFR Part 11
- ICH GCP guidelines
- GDPR and HIPAA requirements
Modern EDC and clinical data management system platforms are designed with compliance at their core, ensuring secure and validated data collection environments.
What to Look For
- System validation and documented controls
- Electronic signatures (eSignatures)
- Role-based access controls (RBAC)
- Built-in compliance reporting
Solutions like Ennov provide validated environments with built-in edit checks and compliance-ready workflows to support data integrity from study start to submission.
Clinical Data Management System Integration with EDC Systems
A clinical data management system should never operate in isolation. Seamless integration with Electronic Data Capture (EDC) systems is essential for efficient data flow and operational visibility.
Why EDC Integration Matters
EDC systems are the primary source of clinical trial data. Integration ensures:
- Real-time data availability
- Reduced manual reconciliation
- Faster database lock
Modern EDC platforms provide centralized access to study data while supporting validation, query management, and reporting.
What to Look For
- Native or tightly integrated EDC functionality
- API-based integrations with third-party systems
- Support for external data sources such as labs, wearables, and ePRO
- Automated data synchronization
Some platforms integrate imaging, lab, and patient-reported data into a single environment, eliminating silos and improving efficiency.
Clinical Data Management System Integration with RIM
Integration between a clinical data management system and Regulatory Information Management (RIM) platforms is becoming increasingly important in 2026.
Why RIM Integration Matters
When integrated with RIM systems:
- Clinical data flows directly into submission-ready formats
- Regulatory teams gain real-time visibility into trial data
- Submission timelines accelerate
Modern RIM platforms support regulatory document management, submission tracking, and compliance oversight.
What to Look For
- Direct integration between clinical and regulatory workflows
- Automated generation of submission-ready datasets such as SDTM
- Shared metadata and document structures
- End-to-end traceability from data capture to submission

Audit Trails in a Clinical Data Management System
Audit trails are essential for compliance, inspections, and long-term data integrity.
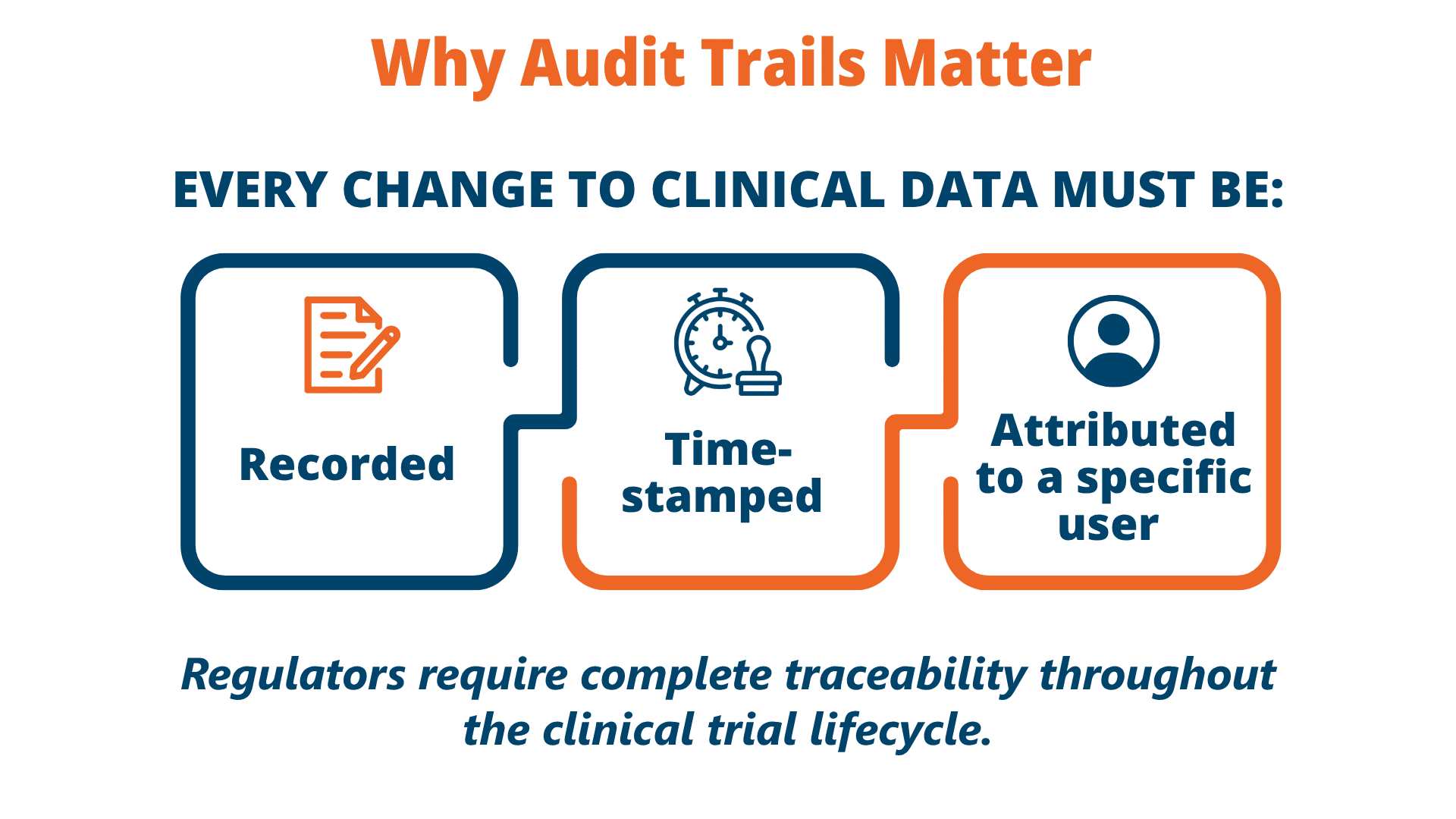
Modern clinical data management system platforms maintain comprehensive audit trails for every action, ensuring transparency and compliance.
What to Look For
- Automatic tracking of all data changes
- User-level activity logs
- Version history and data lineage
- Inspection-ready audit trail reporting
Some systems also allow complete audit trail exports, simplifying inspections and reducing preparation time.
Data Quality and Validation in a Clinical Data Management System
High-quality data is the foundation of successful clinical trials.
Key Features
- Automated edit checks and validation rules
- Query management workflows
- Real-time discrepancy detection
A modern clinical data management system ensures issues are identified early, reducing downstream delays and costly rework.
Multi-Source Integration in a Clinical Data Management System
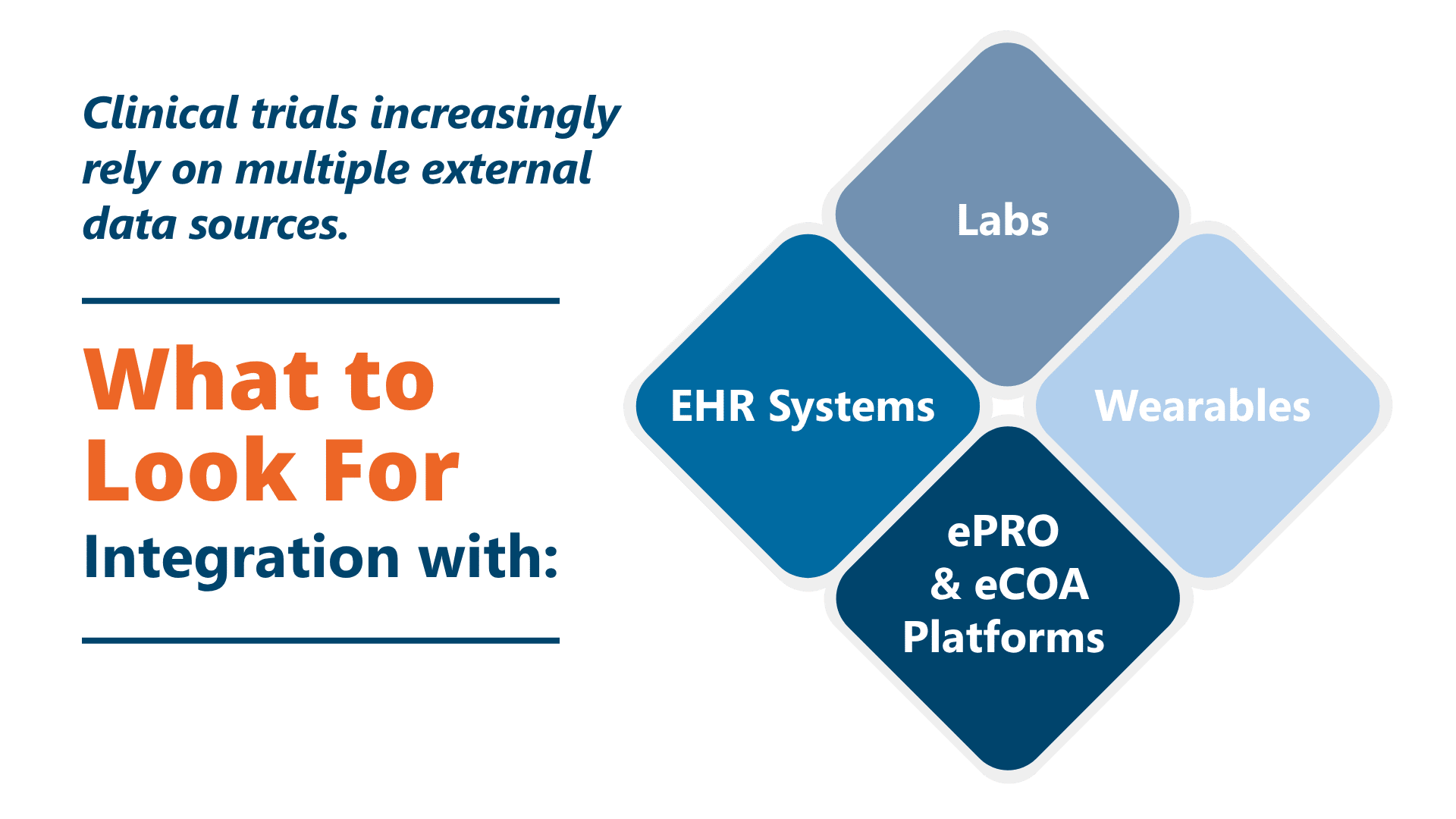
Modern clinical data management system platforms support standards such as HL7, FHIR, and API-based integrations for seamless data ingestion.
Unified Clinical Data Management System Platforms
Fragmented systems create inefficiencies, operational risk, and compliance challenges. The trend in 2026 is moving toward unified clinical platforms.
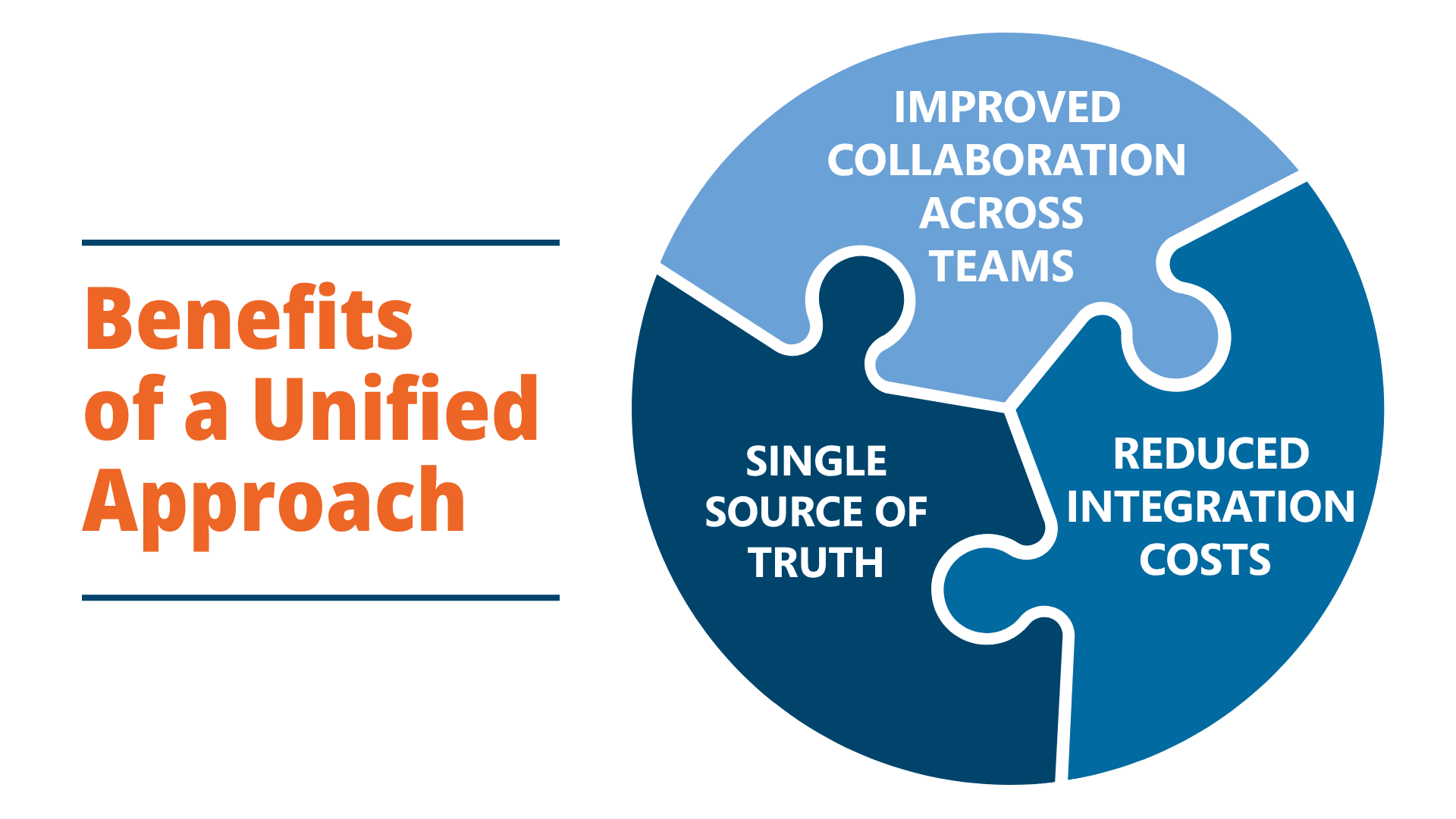
Ennov provides a unified platform that combines:
- CDMS / EDC
- CTMS
- eTMF
- QMS
This connected approach improves visibility and eliminates operational silos across clinical environments.
Scalability and Flexibility in a Clinical Data Management System
Your clinical data management system should scale alongside your clinical pipeline.
What to Look For
- Configurable workflows
- Support for Phase I–IV studies
- Cloud-based deployment
- Global scalability
Flexible systems reduce the need for costly custom development while supporting long-term growth.
User Experience and Adoption
Even the most advanced clinical data management system will fail if users do not adopt it.
Key Considerations
- Intuitive interface
- Minimal training requirements
- Fast study build and deployment
User-friendly systems improve site adoption, operational efficiency, and data quality.
FAQs About Clinical Data Management Systems
What is a clinical data management system?
A clinical data management system is software used to collect, manage, validate, and maintain clinical trial data while ensuring regulatory compliance and data integrity.
Why is a clinical data management system important?
A clinical data management system helps organizations improve data quality, accelerate trial timelines, maintain compliance, and support regulatory submissions.
How does a clinical data management system support compliance?
Modern systems support compliance through audit trails, role-based access controls, electronic signatures, and validated workflows aligned with FDA and ICH guidelines.
What integrations should a clinical data management system support?
A clinical data management system should integrate with EDC, RIM, CTMS, eTMF, labs, ePRO, EHR systems, and external data sources.
Conclusion
Choosing the right clinical data management system is a strategic decision that impacts every stage of a clinical trial.
When evaluating platforms in 2026, prioritize:
- Regulatory compliance
- EDC integration
- RIM integration
- Robust audit trails
- Data integrity and validation
The most effective approach is adopting an integrated platform like Ennov, one that connects clinical data, regulatory processes, and quality management into a single compliant ecosystem.
Learn how Ennov’s clinical data management system supports scalable, compliant, and future-ready clinical operations.
Clinical trials are becoming more data-intensive, regulated, and interconnected. Choosing the right clinical data management system is no longer just about data capture, it’s about ensuring compliance, integration, and traceability across the entire clinical ecosystem.


